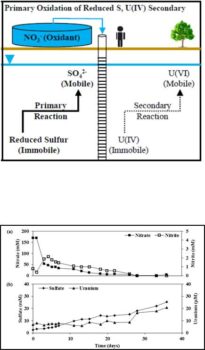
Reoxidation and mobilization of previously reduced and immobilized uranium by dissolved-phase oxidants poses a significant challenge for remediating uranium-contaminated groundwater. Preferential oxidation of reduced sulfur-bearing species, as opposed to reduced uranium-bearing species, has been demonstrated to limit the mobility of uranium at the laboratory scale yet field-scale investigations are lacking. In this study, the mobility of uranium in the presence of nitrate oxidant was investigated in a shallow groundwater system after establishing conditions conducive to uranium reduction and the formation of reduced sulfur-bearing species. A series of three injections of groundwater (200 L) containing U(VI) (5 μM) and amended with ethanol (40 mM) and sulfate (20 mM) were conducted in ten test wells in order to stimulate microbial-mediated reduction of uranium and the formation of reduced sulfur-bearing species. Simultaneous push‐pull tests were then conducted in triplicate well clusters to investigate the mobility of U(VI) under three conditions: 1) high nitrate (120 mM), 2) high nitrate (120 mM) with ethanol (30 mM), and 3) low nitrate (2 mM) with ethanol (30 mM). Dilution-adjusted breakthrough curves of ethanol, nitrate, nitrite, sulfate, and U(VI) suggested that nitrate reduction was predominantly coupled to the oxidation of reduced-sulfur bearing species, as opposed to the reoxidation of U(IV), under all three conditions for the duration of the 36-day tests. The amount of sulfate, but not U(VI), recovered during the push‐pull tests was substantially more than injected, relative to bromide tracer, under all three conditions and further suggested that reduced sulfur-bearing species were preferentially oxidized under nitrate-reducing conditions. However, some reoxidation of U(IV) was observed under nitrate-reducing conditions and in the absence of detectable nitrate and/or nitrite. This suggested that reduced sulfur-bearing species may not be fully effective at limiting the mobility of uranium in the presence of dissolved and/or solid-phase oxidants. The results of this field study confirmed those of previous laboratory studies which suggested that reoxidation of uranium under nitrate-reducing conditions can be substantially limited by preferential oxidation of reduced sulfur-bearing species.
Objective
- Test the in situ mobility of uranium in the presence of nitrate oxidant following uranium- and sulfate-reducing conditions
New Science
- Conducted the first ever field-scale co-injection of ethanol and sulfate to form reduced sulfur-bearing species (S0, FeS, FeS2, MnS) at a uranium-contaminated site
- Subsequent injection of nitrate oxidant and periodic extraction of groundwater demonstrated substantial oxidation of reduced sulfur-bearing species as opposed to reduced/immobilized uranium
- Confirmed previous laboratory studies by demonstrating preferential oxidation of reduced sulfur by nitrate can limit the in situ mobility of uranium
Significance
- Establishing sulfate-reducing conditions following U(VI) reduction can substantially limit the extent of uranium mobility in the presence of nitrate oxidant
