SUMMARY
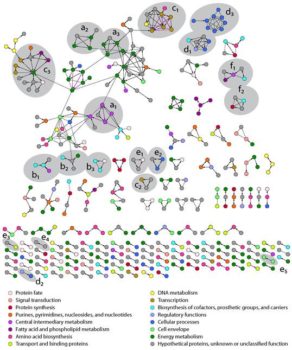
Numerous affinity purification – mass spectrometry (AP-MS) and yeast two-hybrid (Y2H) screens have each defined thousands of pairwise protein-protein interactions (PPIs), most between functionally unrelated proteins. The accuracy of these networks, however, is under debate. Here we present an AP-MS survey of the bacterium Desulfovibrio vulgaris together with a critical reanalysis of nine published bacterial Y2H and AP-MS screens. We have identified 459 high confidence PPIs from D. vulgaris and 391 from Escherichia coli. Compared to the nine published interactomes, our two networks are smaller; are much less highly connected; have significantly lower false discovery rates, and are much more enriched in protein pairs that are encoded in the same operon, have similar functions, and are reproducibly detected in other physical interaction assays. Our work establishes more stringent benchmarks for the properties of protein interactomes and suggests that bona fide PPIs much more frequently involve protein partners that are annotated with similar functions or that can be validated in independent assays than earlier studies suggested.
Mapping the protein interactome of the model sulfate-reducing obligate anaerobe Desulfovibrio vulgaris
- Advance one of ENIGMA’s key missions, evidence-based annotation of gene function, by determining the stable protein interactome of the model sulfate-reducing bacterium Desulfovibrio vulgaris Hildenborough.
New Science
- Conducted high throughput affinity purifications – mass spectrometry study using D. vulgaris
- Used logistic regression to predict a high confidence D. vulgaris interaction network composed of 459 protein-protein interactions (PPIs) at a low 17% false discovery rate
- Established that our two networks are smaller, much less highly connected and have significantly lower false discovery rates than nine published bacterial protein interactomes
- PPIs in our networks are highly enriched for those encoded by the same operon and/or those which share similar annotated functions
Significance
- Established stringent benchmark properties for highly accurate bacterial protein interactomes
